 
Temperature - Imperial Unitsĭensity and specific volume of dry air and water vapor at temperatures ranging 225 to 900 degF (107 to 482 degC).Įlectric heating of an object or mass - temperature change vs. Specific Weight and Specific GravityĪn introduction to density, specific weight and specific gravity.ĭry Air and Water Vapor - Density and Specific Volume vs. Specific volume, enthalpy and entropy of compressed water.ĭensity vs. Phase diagram included.Ĭompressed Water - Properties vs. Temperature and Pressureįigures and table with changes in Prandtl number for carbon dioxide with changes in temperature and pressure.Ĭarbon Dioxide - Thermophysical PropertiesĬhemical, physical and thermal properties of carbon dioxide. 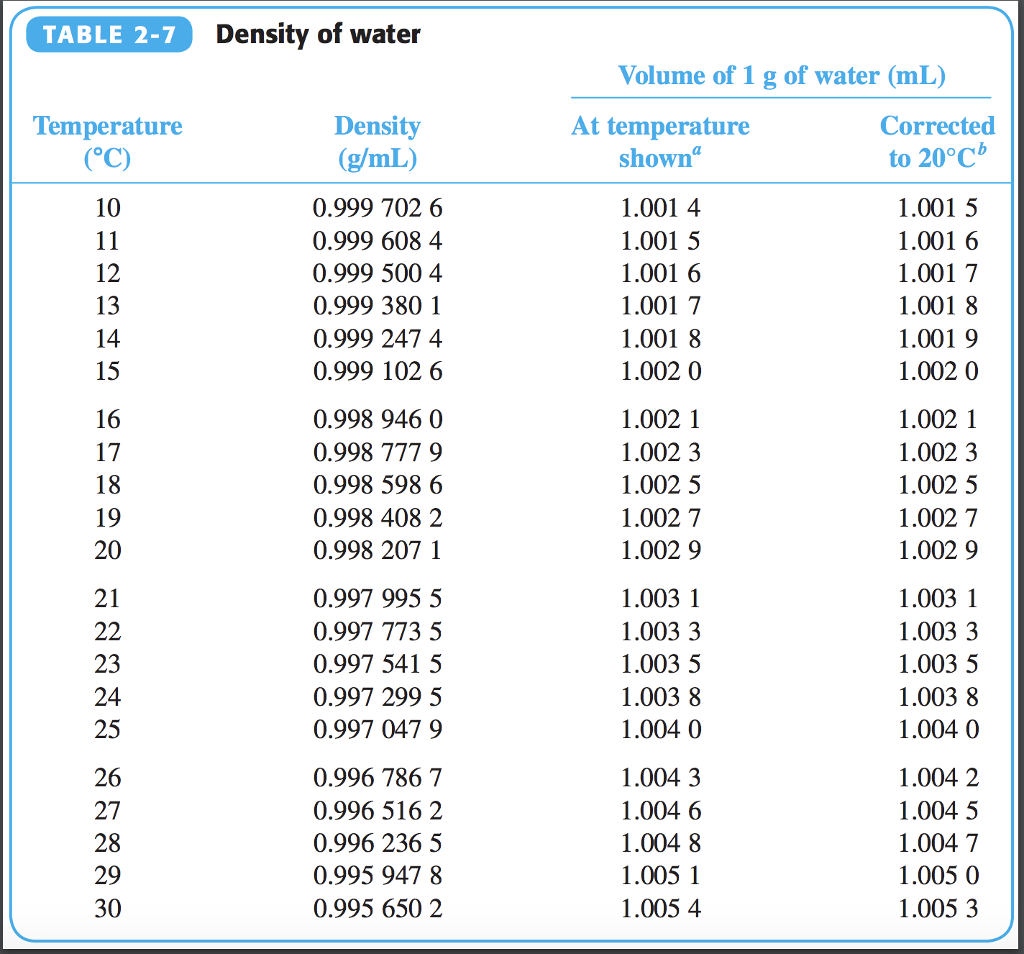
Phase diagram included.Ĭarbon dioxide - Prandtl Number vs. Phase diagram included.Ĭhemical, physical and thermal properties of benzene, also called benzol. Phase diagram included.Ĭhemical, Physical and Thermal Properties of Ammonia. Steam & condensate systems- properties, capacities, pipe sizing, systems configuration and more.Ĭhemical, physical and thermal properties of acetone, also called 2-propanone, dimethyl ketone and pyroacetic acid. Material properties of gases, fluids and solids - densities, specific heats, viscosities and more. Definitions and convertion calculators.Ĭlimate, meteorology, sun, wind and environmental related engineering resources. See also more about atmospheric pressure, and STP - Standard Temperature and Pressure & NTP - Normal Temperature and Pressure ,Īs well as Thermophysical properties of: Acetone, Acetylene, Air, Ammonia, Argon, Benzene, Butane, Carbon dioxide, Carbon monoxide, Ethane, Ethanol, Ethylene, Helium, Hydrogen, Hydrogen sulfide, Methane, Methanol, Nitrogen, Oxygen, Pentane, Propane, Toluene and Heavy water, D 2 O. All properties are given in both SI and imperial units. 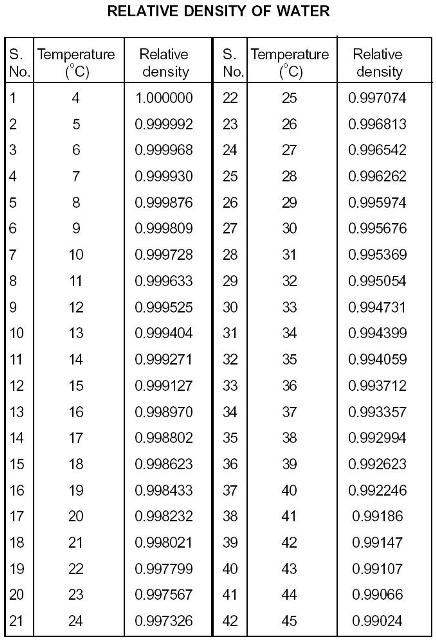
Calculators giving the properties at specified temperatures are also available. Properties at gas-liquid equilibrium conditionįor each topic, there are figures and tables showing the variations in properties with temperature. 
Ionization Constant, pK w, of normal and heavy water.Density, specific weight and thermal expansion coefficient.Triple point temperature: 0.01 ☌ = 32.02 ☏įollow the links below to get values for the listed properties of liquid water at varying pressure and temperature :.Thermal expansion from 4 o C to 100 o C: 4.2x10 -2 (Note! - volumetric temperature expansion of water is not linear with temperature).Specific heat water vapor: 1.996 kJ/kgK =0.4767 Btu(IT)/(lb m ☏) or kcal/(kg K).Latent heat of melting: 334 kJ/kg = 144 Btu(IT)/lb.Bulk modulus elasticity : 2.15 x 10 9 Pa or N/m 2.Here is a video example of how to solve a density problem. Then divide its mass by its volume, remembering to divide the units as well.Ī #"1.26cm"^3# sample of the element mercury has a mass of #"17.05g"#. In order to determine the density of a substance, you need to know its mass and its volume. Volume units are most commonly cubic centimeters ( #"cm"^3#), cubic meters ( #"m"^3#), or millileters (mL).Įxamples of density include the following: Mass units are most commonly grams or kilograms. The density formula is #"density"# = #"mass"/"volume"#. Density measures the compactness in molecular arrangement in any substance which determines how heavy or light any substance is. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
